As per Intent Market Research, the Pharmaceutical Manufacturing Software Market was valued at USD 6.7 Billion in 2024-e and will surpass USD 12.6 Billion by 2030; growing at a CAGR of 11.1% during 2025 - 2030.
The pharmaceutical manufacturing software market plays a crucial role in improving operational efficiency, ensuring compliance, and managing complex processes within the pharmaceutical and biotechnology industries. As the need for more streamlined and optimized production increases, these software solutions provide real-time data, facilitate regulatory compliance, and enhance overall production management. With the industry’s focus on regulatory standards, such as FDA guidelines and Good Manufacturing Practices (GMP), the integration of software like Enterprise Resource Planning (ERP), Manufacturing Execution Systems (MES), and Quality Management Systems (QMS) is becoming essential for managing the entire lifecycle of pharmaceutical production. The market is poised for growth as pharmaceutical companies and contract manufacturers increasingly rely on digital solutions to enhance production efficiency, reduce costs, and improve product quality.
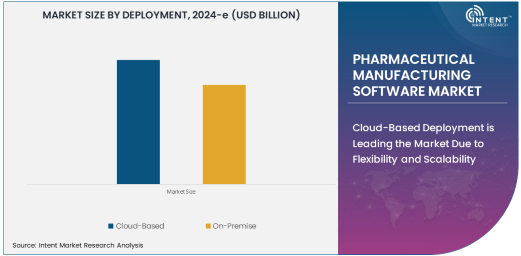
Cloud-Based Deployment is Leading the Market Due to Flexibility and Scalability
The cloud-based deployment model is leading the pharmaceutical manufacturing software market due to its flexibility, scalability, and cost-effectiveness. This deployment method allows pharmaceutical companies to store and manage vast amounts of production data in the cloud, enabling seamless access from various locations and devices. Cloud-based software solutions offer significant advantages, such as reducing the need for on-site IT infrastructure, lowering upfront costs, and providing the ability to scale up or down based on business needs.
Cloud-based software also enhances collaboration between different departments and stakeholders within pharmaceutical companies and contract manufacturing organizations (CMOs). Real-time data sharing and easy integration with other systems streamline communication and decision-making. As the pharmaceutical industry continues to embrace digital transformation, the demand for cloud-based manufacturing software solutions is expected to increase significantly. This technology supports faster implementation and ongoing software updates, helping companies maintain compliance with industry regulations and adapt to changes in manufacturing processes or product lines.
Manufacturing Execution Systems (MES) Are the Fastest Growing Software Type Due to Real-Time Data and Process Optimization
Manufacturing Execution Systems (MES) are the fastest-growing software type in the pharmaceutical manufacturing software market due to their ability to optimize production processes and provide real-time insights into manufacturing operations. MES enables companies to manage and monitor production in real-time, track materials and finished products, and ensure that operations adhere to strict regulatory standards. With MES, pharmaceutical companies can enhance production efficiency, reduce errors, and ensure consistent product quality, all while maintaining compliance with Good Manufacturing Practices (GMP).
As pharmaceutical manufacturing becomes more complex, the need for comprehensive systems that can manage and improve production workflows has grown. MES solutions are particularly valuable in environments that require high precision, such as biologics and sterile drug production. The demand for MES will continue to rise as pharmaceutical companies focus on improving their operational performance and meeting the growing global demand for medications. Furthermore, the integration of MES with other software solutions like Enterprise Resource Planning (ERP) and Quality Management Systems (QMS) allows for even more streamlined production and better decision-making across the entire manufacturing process.
Pharmaceutical Companies Are the Largest End-Use Industry Due to Direct Involvement in Drug Production
Pharmaceutical companies are the largest end-use industry for pharmaceutical manufacturing software due to their direct involvement in the production of drug products. These companies rely heavily on software solutions such as ERP, MES, and QMS to manage their manufacturing operations efficiently, maintain product quality, and ensure compliance with regulatory requirements. Pharmaceutical companies are increasingly adopting integrated software systems to streamline processes from raw material procurement to finished drug packaging.
As regulatory requirements continue to evolve and production demands grow, pharmaceutical companies need to adopt more advanced manufacturing software to stay competitive. By using software to automate routine tasks, track production data, and ensure the safety and efficacy of their products, pharmaceutical companies can enhance their operational efficiency and reduce the risk of errors or recalls. As a result, the pharmaceutical industry remains the dominant end-user of manufacturing software, accounting for the largest share of the market.
North America Is the Largest Region Due to Strong Pharmaceutical Industry Presence and Technological Advancements
North America is the largest region in the pharmaceutical manufacturing software market, driven by its well-established pharmaceutical industry and ongoing technological advancements. The U.S. is home to some of the largest pharmaceutical companies in the world, which are actively adopting advanced manufacturing software solutions to enhance production efficiency and maintain compliance with stringent regulatory standards. In addition, the growing demand for biologics and personalized medicine in North America has led to an increased focus on software solutions that support complex manufacturing processes.
The region's strong IT infrastructure and the increasing adoption of cloud-based software also contribute to its dominance in the market. With a robust regulatory environment and a large number of pharmaceutical manufacturers, North America is expected to maintain its position as the largest market for pharmaceutical manufacturing software in the coming years. The demand for software solutions that can improve manufacturing processes and optimize supply chains will continue to grow, further driving market expansion in this region.

Competitive Landscape and Key Players
The pharmaceutical manufacturing software market is competitive, with several key players providing advanced software solutions to pharmaceutical companies, biotechnology firms, and contract manufacturing organizations (CMOs). Leading companies in this market include SAP, Dassault Systèmes, Oracle, Siemens, Rockwell Automation, and Honeywell, among others. These companies offer a wide range of software solutions such as ERP, MES, LIMS, QMS, and PLM, designed to improve manufacturing efficiency, ensure regulatory compliance, and enhance product quality.
The market is characterized by constant innovation, with companies focusing on integrating advanced technologies like cloud computing, artificial intelligence (AI), and machine learning (ML) into their software solutions to offer better real-time data analytics and predictive capabilities. Additionally, partnerships, collaborations, and mergers and acquisitions are common strategies used by key players to expand their product offerings and strengthen their market position. The competitive landscape continues to evolve as pharmaceutical companies and CMOs seek software solutions that can streamline operations, reduce costs, and improve compliance with ever-changing industry regulations.
Recent Developments:
- SAP SE launched a new cloud-based ERP solution for pharmaceutical manufacturing, offering advanced features for compliance and real-time data management.
- Oracle Corporation introduced a new Manufacturing Execution System (MES) tailored to the needs of pharmaceutical manufacturers to improve production efficiency and traceability.
- Siemens AG partnered with a pharmaceutical manufacturer to implement Industry 4.0 technologies in their production lines, incorporating IoT and automation.
- Rockwell Automation, Inc. expanded its presence in the pharmaceutical market with the launch of a next-generation quality management software, improving compliance and monitoring.
- MasterControl, Inc. announced an update to its QMS software, integrating it with existing pharmaceutical manufacturing systems to streamline production and improve regulatory adherence.
List of Leading Companies:
- SAP SE
- Oracle Corporation
- Dassault Systèmes
- Siemens AG
- Rockwell Automation, Inc.
- Honeywell International Inc.
- Schneider Electric SE
- Aspen Technology, Inc.
- Prolink Solutions
- Limestone Technologies Inc.
- Veeva Systems Inc.
- MasterControl, Inc.
- IQMS (Dassault Systèmes)
- Medidata Solutions
- AbbVie Inc.
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 6.7 Billion |
|
Forecasted Value (2030) |
USD 12.6 Billion |
|
CAGR (2025 – 2030) |
11.1% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Global Pharmaceutical Manufacturing Software Market by Deployment (Cloud-Based, On-Premise), by Software Type (Enterprise Resource Planning (ERP), Manufacturing Execution Systems (MES), Laboratory Information Management Systems (LIMS), Product Lifecycle Management (PLM), Quality Management Systems (QMS)), by End-Use Industry (Pharmaceutical Companies, Biotechnology Companies, Contract Manufacturing Organizations (CMOs), Research & Development (R&D)); Insights & Forecast (2024 – 2030) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
SAP SE, Oracle Corporation, Dassault Systèmes, Siemens AG, Rockwell Automation, Inc., Honeywell International Inc., Aspen Technology, Inc., Prolink Solutions, Limestone Technologies Inc., Veeva Systems Inc., MasterControl, Inc., IQMS (Dassault Systèmes), AbbVie Inc. |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Pharmaceutical Manufacturing Software Market, by Deployment (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Cloud-Based |
|
4.2. On-Premise |
|
5. Pharmaceutical Manufacturing Software Market, by Software Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Enterprise Resource Planning (ERP) |
|
5.2. Manufacturing Execution Systems (MES) |
|
5.3. Laboratory Information Management Systems (LIMS) |
|
5.4. Product Lifecycle Management (PLM) |
|
5.5. Quality Management Systems (QMS) |
|
6. Pharmaceutical Manufacturing Software Market, by End-Use Industry (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Pharmaceutical Companies |
|
6.2. Biotechnology Companies |
|
6.3. Contract Manufacturing Organizations (CMOs) |
|
6.4. Research & Development (R&D) |
|
7. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Regional Overview |
|
7.2. North America |
|
7.2.1. Regional Trends & Growth Drivers |
|
7.2.2. Barriers & Challenges |
|
7.2.3. Opportunities |
|
7.2.4. Factor Impact Analysis |
|
7.2.5. Technology Trends |
|
7.2.6. North America Pharmaceutical Manufacturing Software Market, by Deployment |
|
7.2.7. North America Pharmaceutical Manufacturing Software Market, by Software Type |
|
7.2.8. North America Pharmaceutical Manufacturing Software Market, by End-Use Industry |
|
7.2.9. By Country |
|
7.2.9.1. US |
|
7.2.9.1.1. US Pharmaceutical Manufacturing Software Market, by Deployment |
|
7.2.9.1.2. US Pharmaceutical Manufacturing Software Market, by Software Type |
|
7.2.9.1.3. US Pharmaceutical Manufacturing Software Market, by End-Use Industry |
|
7.2.9.2. Canada |
|
7.2.9.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
7.3. Europe |
|
7.4. Asia-Pacific |
|
7.5. Latin America |
|
7.6. Middle East & Africa |
|
8. Competitive Landscape |
|
8.1. Overview of the Key Players |
|
8.2. Competitive Ecosystem |
|
8.2.1. Level of Fragmentation |
|
8.2.2. Market Consolidation |
|
8.2.3. Product Innovation |
|
8.3. Company Share Analysis |
|
8.4. Company Benchmarking Matrix |
|
8.4.1. Strategic Overview |
|
8.4.2. Product Innovations |
|
8.5. Start-up Ecosystem |
|
8.6. Strategic Competitive Insights/ Customer Imperatives |
|
8.7. ESG Matrix/ Sustainability Matrix |
|
8.8. Manufacturing Network |
|
8.8.1. Locations |
|
8.8.2. Supply Chain and Logistics |
|
8.8.3. Product Flexibility/Customization |
|
8.8.4. Digital Transformation and Connectivity |
|
8.8.5. Environmental and Regulatory Compliance |
|
8.9. Technology Readiness Level Matrix |
|
8.10. Technology Maturity Curve |
|
8.11. Buying Criteria |
|
9. Company Profiles |
|
9.1. SAP SE |
|
9.1.1. Company Overview |
|
9.1.2. Company Financials |
|
9.1.3. Product/Service Portfolio |
|
9.1.4. Recent Developments |
|
9.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
9.2. Oracle Corporation |
|
9.3. Dassault Systèmes |
|
9.4. Siemens AG |
|
9.5. Rockwell Automation, Inc. |
|
9.6. Honeywell International Inc. |
|
9.7. Schneider Electric SE |
|
9.8. Aspen Technology, Inc. |
|
9.9. Prolink Solutions |
|
9.10. Limestone Technologies Inc. |
|
9.11. Veeva Systems Inc. |
|
9.12. MasterControl, Inc. |
|
9.13. IQMS (Dassault Systèmes) |
|
9.14. Medidata Solutions |
|
9.15. AbbVie Inc. |
|
10. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Pharmaceutical Manufacturing Software Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Pharmaceutical Manufacturing Software Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Pharmaceutical Manufacturing Software Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
