As per Intent Market Research, the Pharmaceutical Filtration Market was valued at USD 9.7 Billion in 2024-e and will surpass USD 15.3 Billion by 2030; growing at a CAGR of 7.9% during 2025 - 2030.
The pharmaceutical filtration market plays a pivotal role in ensuring the safety, purity, and quality of drugs during their manufacturing processes. Filtration is crucial in the pharmaceutical industry to remove impurities, bacteria, and other unwanted particles from active pharmaceutical ingredients (APIs), formulations, and final drug products. As the global demand for biopharmaceuticals and generic drugs continues to rise, the need for effective filtration solutions has become more pronounced. The market is driven by stringent regulatory standards, advancements in filtration technology, and the increasing focus on maintaining the highest standards of drug safety and efficacy.
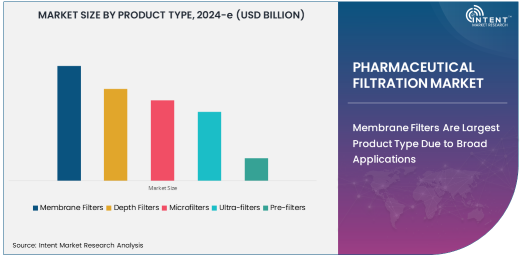
Membrane Filters Are Largest Product Type Due to Broad Applications
Membrane filters dominate the pharmaceutical filtration market, primarily due to their broad applications and high efficiency in separating particles, microorganisms, and contaminants from liquids and gases. These filters are used extensively in both sterile and non-sterile filtration processes, ensuring the removal of microorganisms and particulates to meet stringent pharmaceutical standards.
Membrane filtration technology provides superior performance in applications requiring high levels of sterility, such as in the preparation of injectable drugs, vaccines, and biological products. Its versatility, along with the ability to filter liquids at various pressure levels, makes membrane filters the preferred choice for pharmaceutical manufacturers. The ongoing innovation in membrane materials and technologies is further expanding their use, contributing to their dominance in the market.
Sterile Filtration Is Largest Application Due to Critical Sterility Requirements
Sterile filtration represents the largest application segment in the pharmaceutical filtration market due to the critical importance of maintaining sterility in pharmaceutical products. The growing demand for injectable drugs, biologics, and vaccines, which require absolute sterility, drives the need for high-performance filtration solutions. Sterile filtration ensures the removal of bacteria, endotoxins, and particulate matter, safeguarding the integrity and safety of drug formulations.
As the biopharmaceutical industry expands and the global healthcare sector focuses on the production of biologic therapies and vaccines, the need for sterile filtration continues to rise. Regulatory guidelines also mandate the use of sterile filtration in many pharmaceutical processes, further solidifying its position as the largest application in the market.
Pharmaceutical Manufacturers Are Largest End-Use Industry Due to High Filtration Demand
Pharmaceutical manufacturers represent the largest end-use industry for pharmaceutical filtration solutions. The manufacturing of both traditional and biologic drugs requires effective filtration processes to ensure the removal of contaminants and maintain product quality. As pharmaceutical companies scale up production to meet growing global demand, the need for efficient and reliable filtration systems has become paramount.
In pharmaceutical manufacturing, filtration is critical at various stages of production, from raw material processing to the final drug formulation. The stringent quality standards and regulatory requirements for the production of sterile, non-sterile, and biologic drugs drive the demand for filtration technologies. Pharmaceutical manufacturers rely on advanced filtration systems to meet these standards while maintaining high throughput and minimizing production downtime.
North America Is Largest Region Due to Advanced Healthcare Infrastructure and Regulatory Compliance
North America is the largest region in the pharmaceutical filtration market, driven by advanced healthcare infrastructure, a strong pharmaceutical industry, and strict regulatory compliance requirements. The region is home to some of the world’s largest pharmaceutical companies and contract manufacturing organizations, which significantly contribute to the demand for filtration solutions.
The increasing focus on the production of biologics and biosimilars, along with the rising demand for high-quality medicines, further fuels the growth of the pharmaceutical filtration market in North America. Stringent regulations, such as those from the U.S. Food and Drug Administration (FDA), ensure that filtration processes meet the highest standards of safety and efficacy, further driving the adoption of filtration technologies. The region’s dominance is reinforced by its robust healthcare system and innovative research in the pharmaceutical and biopharmaceutical sectors.

Competitive Landscape and Key Players
The pharmaceutical filtration market is highly competitive, with key players such as Merck KGaA, Sartorius, and Pall Corporation leading the industry. These companies are renowned for providing advanced filtration solutions, including membrane filters, depth filters, and microfilters, for various pharmaceutical applications.
The competitive landscape is characterized by constant innovation and technological advancements, with leading companies investing heavily in R&D to improve the efficiency, capacity, and cost-effectiveness of their filtration products. Additionally, strategic partnerships and acquisitions are common as companies aim to expand their product portfolios and gain access to new markets. As the pharmaceutical industry continues to grow and evolve, these key players remain at the forefront, offering cutting-edge filtration technologies to meet the increasing demand for high-quality drug products.
Recent Developments:
- Merck KGaA expanded its filtration technology portfolio with the launch of a new series of high-performance membrane filters for biopharmaceutical applications.
- Pall Corporation completed a strategic acquisition to enhance its filtration solutions for pharmaceutical manufacturing, particularly in biologics production.
- 3M introduced a new line of advanced filtration membranes designed to meet the growing needs of sterile filtration in the pharmaceutical industry.
- Sartorius AG expanded its bioprocessing capabilities with a new filtration system aimed at improving efficiency and product yield in pharmaceutical manufacturing.
- Donaldson Company, Inc. announced the launch of a next-generation air filtration system specifically designed for pharmaceutical clean rooms, improving air quality and contamination control.
List of Leading Companies:
- Merck KGaA
- Pall Corporation
- Parker Hannifin Corporation
- 3M
- GE Healthcare
- Sartorius AG
- Donaldson Company, Inc.
- Filtration Group Corporation
- MilliporeSigma
- Amazon Filters Ltd.
- Porvair Filtration Group
- Membrane Solutions, LLC
- Lenntech
- Veolia North America
- Mefiag Srl
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 9.7 Billion |
|
Forecasted Value (2030) |
USD 15.3 Billion |
|
CAGR (2025 – 2030) |
7.9% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Global Pharmaceutical Filtration Market by Product Type (Membrane Filters, Depth Filters, Microfilters, Ultra-filters, Pre-filters), by Application (Sterile Filtration, Non-Sterile Filtration), by End-Use Industry (Pharmaceutical Manufacturers, Contract Manufacturing Organizations, Biopharmaceuticals); Insights & Forecast (2024 – 2030) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Merck KGaA, Pall Corporation, Parker Hannifin Corporation, 3M, GE Healthcare, Sartorius AG, Filtration Group Corporation, MilliporeSigma, Amazon Filters Ltd., Porvair Filtration Group, Membrane Solutions, LLC, Lenntech, Mefiag Srl |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Pharmaceutical Filtration Market, by Drug Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Oral Anti-Androgens |
|
4.2. Injectable Anti-Androgens |
|
5. Pharmaceutical Filtration Market, by Indication (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Prostate Cancer |
|
5.2. Polycystic Ovary Syndrome (PCOS) |
|
5.3. Hirsutism |
|
5.4. Other Androgen-Related Disorders |
|
6. Pharmaceutical Filtration Market, by End-Use Industry (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Hospitals and Clinics |
|
6.2. Homecare Settings |
|
6.3. Research and Laboratories |
|
7. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Regional Overview |
|
7.2. North America |
|
7.2.1. Regional Trends & Growth Drivers |
|
7.2.2. Barriers & Challenges |
|
7.2.3. Opportunities |
|
7.2.4. Factor Impact Analysis |
|
7.2.5. Technology Trends |
|
7.2.6. North America Pharmaceutical Filtration Market, by Drug Type |
|
7.2.7. North America Pharmaceutical Filtration Market, by Indication |
|
7.2.8. North America Pharmaceutical Filtration Market, by End-Use Industry |
|
7.2.9. By Country |
|
7.2.9.1. US |
|
7.2.9.1.1. US Pharmaceutical Filtration Market, by Drug Type |
|
7.2.9.1.2. US Pharmaceutical Filtration Market, by Indication |
|
7.2.9.1.3. US Pharmaceutical Filtration Market, by End-Use Industry |
|
7.2.9.2. Canada |
|
7.2.9.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
7.3. Europe |
|
7.4. Asia-Pacific |
|
7.5. Latin America |
|
7.6. Middle East & Africa |
|
8. Competitive Landscape |
|
8.1. Overview of the Key Players |
|
8.2. Competitive Ecosystem |
|
8.2.1. Level of Fragmentation |
|
8.2.2. Market Consolidation |
|
8.2.3. Product Innovation |
|
8.3. Company Share Analysis |
|
8.4. Company Benchmarking Matrix |
|
8.4.1. Strategic Overview |
|
8.4.2. Product Innovations |
|
8.5. Start-up Ecosystem |
|
8.6. Strategic Competitive Insights/ Customer Imperatives |
|
8.7. ESG Matrix/ Sustainability Matrix |
|
8.8. Manufacturing Network |
|
8.8.1. Locations |
|
8.8.2. Supply Chain and Logistics |
|
8.8.3. Product Flexibility/Customization |
|
8.8.4. Digital Transformation and Connectivity |
|
8.8.5. Environmental and Regulatory Compliance |
|
8.9. Technology Readiness Level Matrix |
|
8.10. Technology Maturity Curve |
|
8.11. Buying Criteria |
|
9. Company Profiles |
|
9.1. Astellas Pharma Inc. |
|
9.1.1. Company Overview |
|
9.1.2. Company Financials |
|
9.1.3. Product/Service Portfolio |
|
9.1.4. Recent Developments |
|
9.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
9.2. Bayer AG |
|
9.3. AbbVie Inc. |
|
9.4. Ipsen |
|
9.5. Johnson & Johnson |
|
9.6. Merck & Co., Inc. |
|
9.7. Ferring Pharmaceuticals |
|
9.8. Endo International |
|
9.9. Sun Pharmaceutical Industries Ltd. |
|
9.10. Novartis AG |
|
9.11. Eli Lilly and Company |
|
9.12. Teva Pharmaceutical Industries Ltd. |
|
9.13. Hikma Pharmaceuticals |
|
9.14. Mylan N.V. |
|
9.15. Mundipharma International Limited |
|
10. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Pharmaceutical Filtration Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Pharmaceutical Filtration Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Pharmaceutical Filtration Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
