As per Intent Market Research, the Ornithine Transcarbamylase (OTC) Deficiency Treatment Market was valued at USD 1.7 Billion in 2024-e and will surpass USD 3.2 Billion by 2030; growing at a CAGR of 11.7% during 2025 - 2030.
The Ornithine Transcarbamylase (OTC) deficiency treatment market focuses on therapeutic solutions for managing OTC deficiency, a rare genetic disorder that impairs the liver's ability to detoxify ammonia. This disorder can lead to hyperammonemia, which, if left untreated, may result in neurological damage or even be fatal. The treatment market for OTC deficiency involves medications, dietary supplements, and enzyme replacement therapies, all aimed at controlling ammonia levels and preventing complications. As awareness of OTC deficiency grows, the market for its treatment continues to expand, driven by advancements in medical therapies and the need for more effective and accessible treatment options for patients.
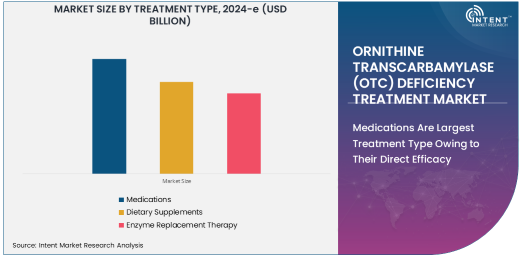
Medications Are Largest Treatment Type Owing to Their Direct Efficacy
Medications represent the largest treatment type in the OTC deficiency treatment market, owing to their direct role in managing ammonia levels in the body. Medications such as ammonia scavengers, which help lower ammonia levels in the blood, are essential for preventing toxic buildup that can damage the brain and other organs. These drugs are commonly used as a part of the treatment regimen to manage the acute and chronic phases of OTC deficiency.
Pharmaceutical interventions are critical in controlling symptoms and preventing life-threatening episodes, making medications the go-to solution for managing OTC deficiency. The availability of effective medications has significantly improved the quality of life for patients with this rare genetic disorder, positioning medications as the largest and most vital treatment segment within the market.
Injectable Route of Administration Is Fastest Growing Owing to Enhanced Efficacy in Acute Cases
Injectable treatments are the fastest-growing route of administration in the OTC deficiency treatment market, primarily due to their rapid action and efficacy in acute cases. Injectable medications, such as intravenous ammonia scavengers, provide a faster and more effective means of managing hyperammonemia, especially in critical situations when oral treatments may not be absorbed quickly enough. This is crucial for patients experiencing acute metabolic crises.
The increasing use of injectable treatments for urgent care in hospitals and specialty clinics is driving the growth of this segment. As advancements in injectable therapies continue to improve treatment outcomes and patient safety, this route of administration is expected to see continued growth in the OTC deficiency treatment market.
Hospitals Are Largest End-Use Industry Owing to Comprehensive Care and Specialized Treatment
Hospitals are the largest end-use industry in the OTC deficiency treatment market, as they provide comprehensive care and specialized treatment for patients with rare genetic disorders. Hospitals are the primary centers for diagnosing and managing OTC deficiency, offering a multidisciplinary approach involving genetic counselors, pediatricians, dietitians, and specialists in metabolic disorders.
Given the complexity of OTC deficiency and the need for prompt and intensive treatment during metabolic crises, hospitals play a crucial role in administering both medications and injectable therapies. The advanced medical facilities and expertise available in hospitals ensure optimal care for patients with OTC deficiency, maintaining hospitals as the largest end-use segment in this market.
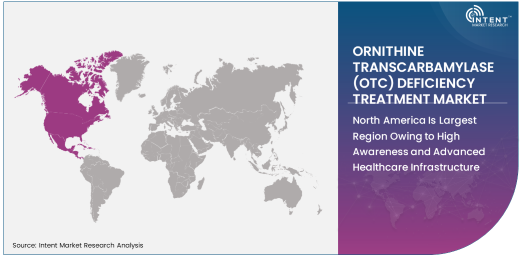
North America Is Largest Region Owing to High Awareness and Advanced Healthcare Infrastructure
North America is the largest region in the OTC deficiency treatment market, driven by the high awareness of rare genetic disorders, advanced healthcare infrastructure, and strong healthcare policy support. In the United States, there are specialized treatment centers for metabolic disorders, and genetic testing is widely available to diagnose OTC deficiency early. Moreover, the region benefits from a robust healthcare system that ensures patients have access to the latest treatments, including medications, dietary supplements, and enzyme replacement therapies.
The high number of patients diagnosed with OTC deficiency in North America, along with increased research and development activities in the region, positions it as the largest market for OTC deficiency treatments. As awareness campaigns and early diagnostic methods continue to improve, North America will maintain its dominance in this market.
Competitive Landscape and Key Players
The OTC deficiency treatment market is competitive, with several pharmaceutical companies leading the development and distribution of treatments. Key players include companies like Hyperion Therapeutics (acquired by Horizon Therapeutics), which provides treatment options for OTC deficiency, as well as companies specializing in enzyme replacement therapies and ammonia scavengers. These players focus on improving treatment efficacy, patient safety, and access to therapies.
The competitive landscape is characterized by ongoing innovation, particularly in the development of enzyme replacement therapies and more effective oral and injectable treatments. Additionally, with the growing awareness of OTC deficiency, new players are entering the market, which is expected to enhance the availability of treatment options and drive future market growth. Key players are investing in research to develop more targeted and personalized therapies, ensuring continued advancements in the treatment of OTC deficiency.
Recent Developments:
- Hyperion Therapeutics, now part of Horizon Therapeutics, expanded its OTC deficiency treatment portfolio by acquiring exclusive rights to a new therapy for urea cycle disorders.
- Biomarin Pharmaceutical Inc. launched a new enzyme replacement therapy for OTC deficiency, significantly improving patient outcomes.
- Ultragenyx Pharmaceutical Inc. announced promising results from a Phase III clinical trial for an OTC deficiency gene therapy.
- Fresenius Kabi AG introduced an injectable OTC deficiency treatment that helps reduce ammonia levels in patients more effectively.
- Sanofi Genzyme acquired ExeGi Pharma, strengthening its position in the OTC deficiency treatment market and enhancing its pipeline for metabolic disorder therapies.
List of Leading Companies:
- Hyperion Therapeutics (acquired by Horizon Therapeutics)
- Fresenius Kabi AG
- Biomarin Pharmaceutical Inc.
- Orphan Europe (Recordati Group)
- Aventis Pharma
- ExeGi Pharma
- Nutricia North America (Danone)
- Ultragenyx Pharmaceutical Inc.
- Horizon Therapeutics
- Shire Pharmaceuticals (now part of Takeda)
- Chiesi Farmaceutici S.p.A.
- Teva Pharmaceutical Industries Ltd.
- Sangamo Therapeutics
- Amgen Inc.
- Sanofi Genzyme
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 1.7 Billion |
|
Forecasted Value (2030) |
USD 3.2 Billion |
|
CAGR (2025 – 2030) |
11.7% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Global Ornithine Transcarbamylase (OTC) Deficiency Treatment Market by Treatment Type (Medications, Dietary Supplements, Enzyme Replacement Therapy), by Route of Administration (Oral, Injectable), by End-Use Industry (Hospitals, Specialty Clinics, Homecare) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Hyperion Therapeutics (acquired by Horizon Therapeutics), Fresenius Kabi AG, Biomarin Pharmaceutical Inc., Orphan Europe (Recordati Group), Aventis Pharma, ExeGi Pharma, Ultragenyx Pharmaceutical Inc., Horizon Therapeutics, Shire Pharmaceuticals (now part of Takeda), Chiesi Farmaceutici S.p.A., Teva Pharmaceutical Industries Ltd., Sangamo Therapeutics, Sanofi Genzyme |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Treatment Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Medications |
|
4.2. Dietary Supplements |
|
4.3. Enzyme Replacement Therapy |
|
5. Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Route of Administration (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Oral |
|
5.2. Injectable |
|
6. Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by End-Use Industry (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Hospitals |
|
6.2. Specialty Clinics |
|
6.3. Homecare |
|
7. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Regional Overview |
|
7.2. North America |
|
7.2.1. Regional Trends & Growth Drivers |
|
7.2.2. Barriers & Challenges |
|
7.2.3. Opportunities |
|
7.2.4. Factor Impact Analysis |
|
7.2.5. Technology Trends |
|
7.2.6. North America Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Treatment Type |
|
7.2.7. North America Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Route of Administration |
|
7.2.8. North America Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by End-Use Industry |
|
7.2.9. By Country |
|
7.2.9.1. US |
|
7.2.9.1.1. US Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Treatment Type |
|
7.2.9.1.2. US Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by Route of Administration |
|
7.2.9.1.3. US Ornithine Transcarbamylase (OTC) Deficiency Treatment Market, by End-Use Industry |
|
7.2.9.2. Canada |
|
7.2.9.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
7.3. Europe |
|
7.4. Asia-Pacific |
|
7.5. Latin America |
|
7.6. Middle East & Africa |
|
8. Competitive Landscape |
|
8.1. Overview of the Key Players |
|
8.2. Competitive Ecosystem |
|
8.2.1. Level of Fragmentation |
|
8.2.2. Market Consolidation |
|
8.2.3. Product Innovation |
|
8.3. Company Share Analysis |
|
8.4. Company Benchmarking Matrix |
|
8.4.1. Strategic Overview |
|
8.4.2. Product Innovations |
|
8.5. Start-up Ecosystem |
|
8.6. Strategic Competitive Insights/ Customer Imperatives |
|
8.7. ESG Matrix/ Sustainability Matrix |
|
8.8. Manufacturing Network |
|
8.8.1. Locations |
|
8.8.2. Supply Chain and Logistics |
|
8.8.3. Product Flexibility/Customization |
|
8.8.4. Digital Transformation and Connectivity |
|
8.8.5. Environmental and Regulatory Compliance |
|
8.9. Technology Readiness Level Matrix |
|
8.10. Technology Maturity Curve |
|
8.11. Buying Criteria |
|
9. Company Profiles |
|
9.1. Hyperion Therapeutics (acquired by Horizon Therapeutics) |
|
9.1.1. Company Overview |
|
9.1.2. Company Financials |
|
9.1.3. Product/Service Portfolio |
|
9.1.4. Recent Developments |
|
9.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
9.2. Fresenius Kabi AG |
|
9.3. Biomarin Pharmaceutical Inc. |
|
9.4. Orphan Europe (Recordati Group) |
|
9.5. Aventis Pharma |
|
9.6. ExeGi Pharma |
|
9.7. Nutricia North America (Danone) |
|
9.8. Ultragenyx Pharmaceutical Inc. |
|
9.9. Horizon Therapeutics |
|
9.10. Shire Pharmaceuticals (now part of Takeda) |
|
9.11. Chiesi Farmaceutici S.p.A. |
|
9.12. Teva Pharmaceutical Industries Ltd. |
|
9.13. Sangamo Therapeutics |
|
9.14. Amgen Inc. |
|
9.15. Sanofi Genzyme |
|
10. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Ornithine Transcarbamylase (OTC) Deficiency Treatment Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Ornithine Transcarbamylase (OTC) Deficiency Treatment Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Ornithine Transcarbamylase (OTC) Deficiency Treatment Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
