As per Intent Market Research, the Opioid Induced Constipation Treatment Market was valued at USD 128.8 million in 2024-e and will surpass USD 201.6 million by 2030; growing at a CAGR of 7.7% during 2025 - 2030.
The opioid-induced constipation (OIC) treatment market addresses a significant challenge faced by individuals on opioid therapy. Opioids, while effective for pain management, can cause constipation as a common side effect, leading to discomfort and further health complications for patients. As opioid use continues to rise globally, particularly for chronic pain conditions, the prevalence of opioid-induced constipation also increases. This has driven the demand for effective treatments to manage and alleviate the symptoms of OIC, contributing to the growth of this market.
The OIC treatment market is characterized by a variety of treatment types, including prescription medications, over-the-counter (OTC) solutions, and injectable medications. Different drug classes, such as peripherally acting mu-opioid receptor antagonists (PAMORAs), laxatives, and prokinetic agents, are employed to relieve the symptoms of constipation. The market is further segmented by application, focusing on acute, chronic, and preventive treatments, while end-users primarily include hospitals, outpatient clinics, and home care settings. As awareness of OIC grows and new treatments are developed, the market for opioid-induced constipation treatment is expected to expand.
Prescription Medications Segment Dominates the Market Owing to Efficacy in Severe Cases
The prescription medications segment holds the largest share in the opioid-induced constipation treatment market. These medications are typically used to manage severe cases of OIC where over-the-counter solutions may not be effective. Prescription treatments include PAMORAs, such as methylnaltrexone and naloxegol, which target the opioid receptors in the gastrointestinal tract to alleviate constipation without affecting the pain-relieving effects of opioids.
Given the potency and targeted action of prescription medications, they are preferred in cases of chronic or severe OIC, particularly in patients receiving long-term opioid therapy for conditions like cancer or chronic pain disorders. As these medications offer significant relief from the discomfort of constipation while minimizing the risk of opioid withdrawal, their adoption is expected to continue to grow, ensuring the dominance of the prescription medications segment in the market.
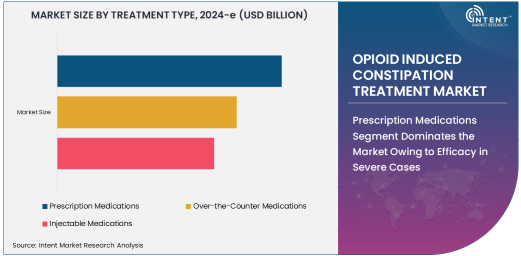
Over-the-Counter Medications Segment Shows Rapid Growth Owing to Accessibility and Affordability
The over-the-counter (OTC) medications segment is witnessing rapid growth, driven by the increasing demand for easily accessible and affordable solutions to manage OIC. OTC medications, such as fiber supplements, stimulant laxatives, and osmotic agents, provide a convenient option for patients who experience mild to moderate OIC. These medications are often the first line of treatment for individuals who prefer non-prescription solutions or are experiencing temporary symptoms of constipation.
The OTC segment benefits from a growing awareness of OIC and the availability of self-administered treatments that do not require a doctor’s prescription. As healthcare professionals recommend the use of OTC treatments for mild cases, the demand for these medications is expected to rise, particularly in the outpatient and home care settings.
Peripherally Acting Mu-Opioid Receptor Antagonists (PAMORAs) Drug Class Leads the Market Owing to Targeted Action
The peripherally acting mu-opioid receptor antagonists (PAMORAs) drug class is the leading therapeutic class in the OIC treatment market, owing to its targeted action on opioid receptors in the gastrointestinal tract. Unlike traditional laxatives, which work by stimulating the entire digestive system, PAMORAs specifically block the opioid receptors in the intestines, improving bowel movement without interfering with the pain-relieving effects of opioids.
PAMORAs are particularly effective in managing chronic OIC, which is often seen in patients on long-term opioid therapy. The ability to manage constipation effectively without altering the opioid’s efficacy in pain relief has made PAMORAs the treatment of choice in severe cases of OIC, contributing to the growth of this drug class in the market.
Chronic Opioid-Induced Constipation Application Leads the Market Due to Long-Term Use of Opioids
The chronic opioid-induced constipation application dominates the market, driven by the long-term use of opioids in managing chronic pain conditions. Chronic pain disorders, such as cancer pain, neuropathy, and arthritis, require prolonged opioid use, which often results in persistent OIC. As such, the treatment of chronic OIC is a major focus within the market.
Given the need for long-term management, chronic OIC often requires a combination of pharmacological treatments, lifestyle modifications, and regular monitoring. The demand for effective, long-term solutions to chronic OIC has led to the dominance of this application, with prescription medications, particularly PAMORAs, playing a central role in managing the condition.
Preventive Treatment Segment Shows Steady Growth Owing to Increased Awareness and Early Intervention
The preventive treatment segment is showing steady growth, owing to the increased awareness of OIC and the benefits of early intervention. Preventive treatments, which include the use of laxatives and bowel-regulating agents at the onset of opioid therapy, are increasingly being recommended by healthcare providers to mitigate the risk of OIC.
By addressing constipation proactively, patients are able to avoid the more severe symptoms of OIC, reducing the need for more intensive treatments later on. The growing recognition of the importance of preventive care in opioid therapy, especially in patients starting long-term opioid use, is driving the growth of this segment.
Hospitals End-User Segment Holds the Largest Share Owing to High Volume of Patients on Opioid Therapy
The hospitals end-user segment holds the largest share in the opioid-induced constipation treatment market. Hospitals are the primary setting for patients who are undergoing major surgeries, receiving cancer treatments, or being treated for chronic pain conditions that necessitate the use of opioids. As these patients are at high risk of developing OIC, hospitals play a central role in the diagnosis and treatment of the condition.
Given the high volume of patients requiring opioid therapy, hospitals are the largest consumers of OIC treatments, particularly prescription medications and injectable treatments. The hospital environment also allows for more intensive monitoring and management of OIC, ensuring that patients receive appropriate treatment to relieve symptoms and prevent complications.
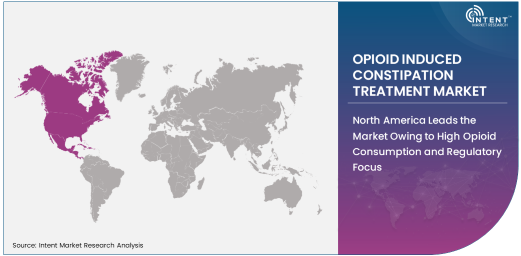
North America Leads the Market Owing to High Opioid Consumption and Regulatory Focus
North America is the largest market for opioid-induced constipation treatments, driven by the high levels of opioid consumption in the United States and Canada. The widespread use of opioids for pain management, particularly in chronic pain and cancer care, has led to a significant prevalence of OIC in the region.
Furthermore, the regulatory focus on opioid use in North America, particularly in response to the opioid crisis, has increased awareness of OIC and the need for effective treatments. As healthcare providers and patients become more proactive in managing OIC, the demand for specialized treatments in North America is expected to continue to grow.
Competitive Landscape and Key Players
The opioid-induced constipation treatment market is competitive, with several key players leading the way, including Ironwood Pharmaceuticals, Shire (now part of Takeda), Valeant Pharmaceuticals (now Bausch Health), and Mylan Pharmaceuticals. These companies are at the forefront of developing and marketing treatments for OIC, particularly PAMORAs and other prescription medications.
With the ongoing opioid epidemic and the increasing recognition of OIC as a significant side effect of opioid therapy, these companies are focused on developing innovative solutions to meet the growing demand for effective treatments. Research and development efforts are focused on creating new drug formulations, improving patient outcomes, and addressing the challenges of opioid dependency and abuse.
Recent Developments:
- In December 2024, Takeda Pharmaceuticals launched a new PAMORA-based treatment for opioid-induced constipation with improved patient compliance.
- In November 2024, Bristol-Myers Squibb received FDA approval for its new prokinetic agent designed for chronic OIC management.
- In October 2024, Valeant Pharmaceuticals announced the release of a next-generation laxative formula aimed at opioid-induced constipation in outpatient settings.
- In September 2024, Shire Pharmaceuticals, under Takeda, introduced a combination therapy targeting OIC with opioid pain relief.
- In August 2024, Ironwood Pharmaceuticals expanded its portfolio with an innovative oral treatment for opioid-induced constipation to improve digestive function.
List of Leading Companies:
- Shire Pharmaceuticals (Acquired by Takeda)
- Valeant Pharmaceuticals (Bausch Health)
- Progenics Pharmaceuticals (Acquired by Lantheus Medical Imaging)
- Bristol-Myers Squibb
- Pfizer Inc.
- Takeda Pharmaceuticals
- McNeil Consumer Healthcare (Johnson & Johnson)
- Ironwood Pharmaceuticals
- Allergan (Acquired by AbbVie)
- Mylan N.V.
- AbbVie Inc.
- AstraZeneca
- Novo Nordisk
- Salix Pharmaceuticals (A Division of Valeant)
- Mallinckrodt Pharmaceuticals
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 128.8 million |
|
Forecasted Value (2030) |
USD 201.6 million |
|
CAGR (2025 – 2030) |
7.7% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Opioid Induced Constipation Treatment Market By Treatment Type (Prescription Medications, Over-the-Counter Medications, Injectable Medications), By Drug Class (Peripherally Acting Mu-Opioid Receptor Antagonists (PAMORAs), Laxatives, Prokinetic Agents), By Application (Acute Opioid-Induced Constipation, Chronic Opioid-Induced Constipation, Preventive Treatment), By End-User (Hospitals, Outpatient Clinics, Home Care) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Shire Pharmaceuticals (Acquired by Takeda), Valeant Pharmaceuticals (Bausch Health), Progenics Pharmaceuticals (Acquired by Lantheus Medical Imaging), Bristol-Myers Squibb, Pfizer Inc., Takeda Pharmaceuticals, McNeil Consumer Healthcare (Johnson & Johnson), Ironwood Pharmaceuticals, Allergan (Acquired by AbbVie), Mylan N.V., AbbVie Inc., AstraZeneca, Novo Nordisk, Salix Pharmaceuticals (A Division of Valeant), Mallinckrodt Pharmaceuticals |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Opioid Induced Constipation Treatment Market, by Treatment Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Prescription Medications |
|
4.2. Over-the-Counter Medications |
|
4.3. Injectable Medications |
|
5. Opioid Induced Constipation Treatment Market, by Drug Class (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Peripherally Acting Mu-Opioid Receptor Antagonists (PAMORAs) |
|
5.2. Laxatives |
|
5.3. Prokinetic Agents |
|
6. Opioid Induced Constipation Treatment Market, by Application (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Acute Opioid-Induced Constipation |
|
6.2. Chronic Opioid-Induced Constipation |
|
6.3. Preventive Treatment |
|
7. Opioid Induced Constipation Treatment Market, by End-User (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Hospitals |
|
7.2. Outpatient Clinics |
|
7.3. Home Care |
|
8. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
8.1. Regional Overview |
|
8.2. North America |
|
8.2.1. Regional Trends & Growth Drivers |
|
8.2.2. Barriers & Challenges |
|
8.2.3. Opportunities |
|
8.2.4. Factor Impact Analysis |
|
8.2.5. Technology Trends |
|
8.2.6. North America Opioid Induced Constipation Treatment Market, by Treatment Type |
|
8.2.7. North America Opioid Induced Constipation Treatment Market, by Drug Class |
|
8.2.8. North America Opioid Induced Constipation Treatment Market, by Application |
|
8.2.9. North America Opioid Induced Constipation Treatment Market, by End-User |
|
8.2.10. By Country |
|
8.2.10.1. US |
|
8.2.10.1.1. US Opioid Induced Constipation Treatment Market, by Treatment Type |
|
8.2.10.1.2. US Opioid Induced Constipation Treatment Market, by Drug Class |
|
8.2.10.1.3. US Opioid Induced Constipation Treatment Market, by Application |
|
8.2.10.1.4. US Opioid Induced Constipation Treatment Market, by End-User |
|
8.2.10.2. Canada |
|
8.2.10.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
8.3. Europe |
|
8.4. Asia-Pacific |
|
8.5. Latin America |
|
8.6. Middle East & Africa |
|
9. Competitive Landscape |
|
9.1. Overview of the Key Players |
|
9.2. Competitive Ecosystem |
|
9.2.1. Level of Fragmentation |
|
9.2.2. Market Consolidation |
|
9.2.3. Product Innovation |
|
9.3. Company Share Analysis |
|
9.4. Company Benchmarking Matrix |
|
9.4.1. Strategic Overview |
|
9.4.2. Product Innovations |
|
9.5. Start-up Ecosystem |
|
9.6. Strategic Competitive Insights/ Customer Imperatives |
|
9.7. ESG Matrix/ Sustainability Matrix |
|
9.8. Manufacturing Network |
|
9.8.1. Locations |
|
9.8.2. Supply Chain and Logistics |
|
9.8.3. Product Flexibility/Customization |
|
9.8.4. Digital Transformation and Connectivity |
|
9.8.5. Environmental and Regulatory Compliance |
|
9.9. Technology Readiness Level Matrix |
|
9.10. Technology Maturity Curve |
|
9.11. Buying Criteria |
|
10. Company Profiles |
|
10.1. Shire Pharmaceuticals (Acquired by Takeda) |
|
10.1.1. Company Overview |
|
10.1.2. Company Financials |
|
10.1.3. Product/Service Portfolio |
|
10.1.4. Recent Developments |
|
10.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
10.2. Valeant Pharmaceuticals (Bausch Health) |
|
10.3. Progenics Pharmaceuticals (Acquired by Lantheus Medical Imaging) |
|
10.4. Bristol-Myers Squibb |
|
10.5. Pfizer Inc. |
|
10.6. Takeda Pharmaceuticals |
|
10.7. McNeil Consumer Healthcare (Johnson & Johnson) |
|
10.8. Ironwood Pharmaceuticals |
|
10.9. Allergan (Acquired by AbbVie) |
|
10.10. Mylan N.V. |
|
10.11. AbbVie Inc. |
|
10.12. AstraZeneca |
|
10.13. Novo Nordisk |
|
10.14. Salix Pharmaceuticals (A Division of Valeant) |
|
10.15. Mallinckrodt Pharmaceuticals |
|
11. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Opioid Induced Constipation Treatment Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Opioid Induced Constipation Treatment Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Opioid Induced Constipation Treatment Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
