As per Intent Market Research, the Lyme Disease Diagnostics Market was valued at USD 1.7 Billion in 2024-e and will surpass USD 2.1 Billion by 2030; growing at a CAGR of 3.8% during 2025-2030.
The Lyme disease diagnostics market is experiencing significant growth due to increased awareness, rising incidences of Lyme disease, and advancements in diagnostic technologies. Lyme disease, caused by the bacterium Borrelia burgdorferi, is transmitted through tick bites, and its early detection is critical for effective treatment. The market is primarily driven by the ongoing research efforts to enhance diagnostic accuracy and reduce the time required for diagnosis. Additionally, improved access to diagnostic tools in endemic regions is further fueling market expansion. This market encompasses a variety of diagnostic methods, including immunoassay tests, PCR tests, microscopy-based tests, and lateral flow assays, each offering unique advantages depending on the stage of infection.
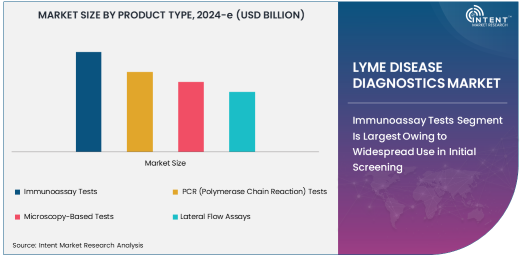
Immunoassay Tests Segment Is Largest Owing to Widespread Use in Initial Screening
Immunoassay tests are currently the largest segment in the Lyme disease diagnostics market. These tests are widely used as the first line of defense in diagnosing Lyme disease due to their simplicity, affordability, and high throughput in clinical settings. Immunoassay tests, such as enzyme-linked immunosorbent assays (ELISA), detect antibodies produced in response to Borrelia infection. These tests offer the benefit of detecting the disease in its early stages, when antibody levels are relatively low but still detectable. The growing demand for rapid, high-throughput diagnostic solutions in both hospitals and diagnostic laboratories is expected to continue driving the immunoassay segment’s dominance in the market.
Immunoassay tests are especially preferred in areas with high tick exposure, where early detection is crucial to prevent complications and severe disease progression. As healthcare systems globally focus on providing faster and more efficient diagnostic tools, the reliance on immunoassay tests for Lyme disease detection will continue to increase. Additionally, advancements in immunoassay technology are enhancing their specificity and sensitivity, further solidifying their place as the most commonly used diagnostic tool in Lyme disease diagnosis.
PCR Tests Segment Is Fastest Growing Owing to High Accuracy and Sensitivity
PCR (Polymerase Chain Reaction) tests are the fastest growing segment within the Lyme disease diagnostics market. These molecular tests offer unmatched accuracy, sensitivity, and specificity, especially in detecting Lyme disease in its early stages, even before the body has developed detectable antibody responses. PCR tests work by amplifying the DNA of Borrelia bacteria, making it detectable even in low concentrations. As a result, PCR tests are increasingly being used in clinical settings to confirm the presence of the disease, particularly in cases where immunoassay results are inconclusive.
The growing adoption of PCR technology in Lyme disease diagnosis is driven by the increasing demand for precise and reliable diagnostic methods. Moreover, the ability of PCR tests to detect the disease in its early stages, even in patients with atypical symptoms, is expanding their use across various healthcare settings. As research into Lyme disease diagnostics continues to advance, the PCR segment is expected to witness substantial growth, with the technology playing a pivotal role in improving diagnostic outcomes.
Enzyme-Linked Immunosorbent Assay (ELISA) Technology Is Largest Owing to Popularity and Accessibility
Enzyme-Linked Immunosorbent Assay (ELISA) is the largest technology used in Lyme disease diagnostics due to its wide availability, ease of use, and cost-effectiveness. ELISA is a popular immunoassay technique used for detecting antibodies specific to Borrelia bacteria in the patient's blood. It is particularly useful in diagnosing Lyme disease in its early stages, offering a high level of sensitivity and specificity. The assay involves the use of an enzyme-labeled antibody to detect the presence of the Lyme bacteria-specific antibodies in the blood, which is a critical early indicator of infection.
The widespread adoption of ELISA across hospitals and diagnostic laboratories is a key factor contributing to its dominance in the Lyme disease diagnostics market. ELISA tests are relatively inexpensive, making them accessible to a broad range of healthcare facilities, including those in resource-constrained environments. Furthermore, advancements in ELISA technology are improving diagnostic accuracy, solidifying its position as the leading diagnostic method for Lyme disease detection.
Hospitals Segment Is Largest Owing to High Diagnostic Demand
Hospitals are the largest end-user segment in the Lyme disease diagnostics market. Hospitals are key centers for diagnosing and treating Lyme disease due to their advanced facilities, specialist staff, and access to cutting-edge diagnostic technologies. The growing number of Lyme disease cases, particularly in endemic regions, has led to an increased demand for rapid and accurate diagnostic solutions in hospital settings. Hospitals rely on a range of diagnostic tests, including immunoassays, PCR tests, and ELISA, to ensure timely and accurate Lyme disease diagnosis.
In addition to the increasing prevalence of Lyme disease, hospitals are also adopting advanced diagnostic technologies to improve patient outcomes. The availability of specialized diagnostic laboratories within hospitals ensures that patients receive timely treatment after a swift and accurate diagnosis. With healthcare systems focusing more on early detection and proactive treatment, the hospital segment is expected to remain the largest contributor to the Lyme disease diagnostics market.
North America Is Largest Region Owing to High Incidence of Lyme Disease
North America is the largest region in the Lyme disease diagnostics market, driven by the high incidence of Lyme disease, particularly in the United States and Canada. The region has seen a significant rise in Lyme disease cases in recent years, primarily due to changes in climate and the expansion of tick populations. The U.S. in particular is a leading market for Lyme disease diagnostics, with extensive healthcare infrastructure and a high demand for advanced diagnostic tools. The widespread implementation of Lyme disease testing in both public health campaigns and clinical practices further boosts the market in this region.
The high awareness levels regarding Lyme disease in North America, coupled with government initiatives aimed at improving disease detection and treatment, contribute to the region's dominance in the global market. As a result, North America is expected to continue leading the Lyme disease diagnostics market, accounting for a significant share of the global revenue.
Competitive Landscape and Key Players
The Lyme disease diagnostics market is highly competitive, with key players focusing on product innovation, mergers and acquisitions, and strategic partnerships to strengthen their market positions. Leading companies in this market include Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers, Hologic Inc., Bio-Rad Laboratories, and Cepheid (Danaher Corporation), among others. These companies are investing in advanced research to develop more accurate, faster, and cost-effective diagnostic tests for Lyme disease.
The competitive landscape is characterized by a mix of established players with significant market shares and emerging companies introducing innovative diagnostic solutions. Companies are increasingly looking to expand their product portfolios and reach by acquiring smaller firms with complementary technologies. As the market evolves, partnerships between diagnostic solution providers and healthcare institutions are expected to drive further growth, particularly in regions with high incidences of Lyme disease.
Recent Developments:
- Abbott Laboratories launched a new PCR-based Lyme disease diagnostic test in 2024, designed to provide faster and more accurate results.
- Thermo Fisher Scientific acquired a molecular diagnostics company in 2023 to expand its Lyme disease testing capabilities and product portfolio.
- Hologic Inc. received FDA approval for a new Lyme disease immunoassay test in late 2023, significantly improving diagnostic accuracy.
- Cepheid introduced a rapid PCR diagnostic test for Lyme disease in 2024, aimed at reducing test turnaround time in clinical settings.
- Bio-Rad Laboratories expanded its Lyme disease diagnostic offerings by adding multiplex PCR kits to its portfolio in 2023 to enable more comprehensive testing.
List of Leading Companies:
- Abbott Laboratories
- Thermo Fisher Scientific
- Siemens Healthineers
- Hologic Inc.
- Bio-Rad Laboratories
- Cepheid (Danaher Corporation)
- Quidel Corporation
- MedMira Inc.
- F. Hoffmann-La Roche Ltd.
- Ortho Clinical Diagnostics
- Becton, Dickinson and Company (BD)
- Armin Laboratories
- Vir Biotechnology
- DiaSorin S.p.A.
- Eppendorf AG
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 1.7 Billion |
|
Forecasted Value (2030) |
USD 2.1 Billion |
|
CAGR (2025 – 2030) |
3.8% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Lyme Disease Diagnostics Market By Product Type (Immunoassay Tests, PCR Tests, Microscopy-Based Tests, Lateral Flow Assays), By Technology (Enzyme-Linked Immunosorbent Assay, Western Blot, PCR, Immunofluorescence Assay, Lateral Flow Immunoassays), By End-User (Hospitals, Diagnostic Laboratories, Research Institutes) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers, Hologic Inc., Bio-Rad Laboratories, Cepheid (Danaher Corporation), Quidel Corporation, MedMira Inc., F. Hoffmann-La Roche Ltd., Ortho Clinical Diagnostics, Becton, Dickinson and Company (BD), Armin Laboratories, Vir Biotechnology, DiaSorin S.p.A., Eppendorf AG |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Lyme Disease Diagnostics Market, by Product Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Immunoassay Tests |
|
4.2. PCR (Polymerase Chain Reaction) Tests |
|
4.3. Microscopy-Based Tests |
|
4.4. Lateral Flow Assays |
|
5. Lyme Disease Diagnostics Market, by Technology (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Enzyme-Linked Immunosorbent Assay (ELISA) |
|
5.2. Western Blot |
|
5.3. PCR |
|
5.4. Immunofluorescence Assay (IFA) |
|
5.5. Lateral Flow Immunoassays |
|
6. Lyme Disease Diagnostics Market, by End-User (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Hospitals |
|
6.2. Diagnostic Laboratories |
|
6.3. Research Institutes |
|
7. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Regional Overview |
|
7.2. North America |
|
7.2.1. Regional Trends & Growth Drivers |
|
7.2.2. Barriers & Challenges |
|
7.2.3. Opportunities |
|
7.2.4. Factor Impact Analysis |
|
7.2.5. Technology Trends |
|
7.2.6. North America Lyme Disease Diagnostics Market, by Product Type |
|
7.2.7. North America Lyme Disease Diagnostics Market, by Technology |
|
7.2.8. North America Lyme Disease Diagnostics Market, by End-User |
|
7.2.9. By Country |
|
7.2.9.1. US |
|
7.2.9.1.1. US Lyme Disease Diagnostics Market, by Product Type |
|
7.2.9.1.2. US Lyme Disease Diagnostics Market, by Technology |
|
7.2.9.1.3. US Lyme Disease Diagnostics Market, by End-User |
|
7.2.9.2. Canada |
|
7.2.9.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
7.3. Europe |
|
7.4. Asia-Pacific |
|
7.5. Latin America |
|
7.6. Middle East & Africa |
|
8. Competitive Landscape |
|
8.1. Overview of the Key Players |
|
8.2. Competitive Ecosystem |
|
8.2.1. Level of Fragmentation |
|
8.2.2. Market Consolidation |
|
8.2.3. Product Innovation |
|
8.3. Company Share Analysis |
|
8.4. Company Benchmarking Matrix |
|
8.4.1. Strategic Overview |
|
8.4.2. Product Innovations |
|
8.5. Start-up Ecosystem |
|
8.6. Strategic Competitive Insights/ Customer Imperatives |
|
8.7. ESG Matrix/ Sustainability Matrix |
|
8.8. Manufacturing Network |
|
8.8.1. Locations |
|
8.8.2. Supply Chain and Logistics |
|
8.8.3. Product Flexibility/Customization |
|
8.8.4. Digital Transformation and Connectivity |
|
8.8.5. Environmental and Regulatory Compliance |
|
8.9. Technology Readiness Level Matrix |
|
8.10. Technology Maturity Curve |
|
8.11. Buying Criteria |
|
9. Company Profiles |
|
9.1. Abbott Laboratories |
|
9.1.1. Company Overview |
|
9.1.2. Company Financials |
|
9.1.3. Product/Service Portfolio |
|
9.1.4. Recent Developments |
|
9.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
9.2. Thermo Fisher Scientific |
|
9.3. Siemens Healthineers |
|
9.4. Hologic Inc. |
|
9.5. Bio-Rad Laboratories |
|
9.6. Cepheid (Danaher Corporation) |
|
9.7. Quidel Corporation |
|
9.8. MedMira Inc. |
|
9.9. F. Hoffmann-La Roche Ltd. |
|
9.10. Ortho Clinical Diagnostics |
|
9.11. Becton, Dickinson and Company (BD) |
|
9.12. Armin Laboratories |
|
9.13. Vir Biotechnology |
|
9.14. DiaSorin S.p.A. |
|
9.15. Eppendorf AG |
|
10. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Lyme Disease Diagnostics Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Lyme Disease Diagnostics Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Lyme Disease Diagnostics Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
