As per Intent Market Research, the Oral Typhoid Vaccine Market was valued at USD 0.4 Billion in 2024-e and will surpass USD 0.7 Billion by 2030; growing at a CAGR of 12.2% during 2025 - 2030.
The Oral Typhoid Vaccine Market is experiencing growth due to the increasing global awareness of typhoid fever and the rising demand for accessible vaccination methods. Typhoid fever, caused by the bacterium Salmonella typhi, remains a significant health issue in many developing countries, where sanitation and clean water access are limited. Oral vaccines, offering a convenient and non-invasive alternative to traditional injectable vaccines, are gaining traction due to their ease of administration and cost-effectiveness. The market is driven by the need to reduce the burden of typhoid fever through vaccination, especially in regions where the disease is endemic, and to provide solutions that are simple for healthcare systems to implement.
Live Attenuated Oral Typhoid Vaccine Segment Is Largest Owing to Efficacy and Long-Term Protection
The live attenuated oral typhoid vaccine segment is the largest in the market, primarily due to its proven efficacy and ability to offer long-term protection against typhoid fever. Live attenuated vaccines are made from weakened forms of the Salmonella typhi bacterium, which stimulates an immune response without causing the disease. This vaccine type has been shown to provide robust protection and has a long shelf life, making it particularly suitable for large-scale vaccination campaigns in endemic regions.
The convenience of the oral form further drives its widespread use, as it eliminates the need for needles and syringes, making it more acceptable and easier to distribute. The availability of oral vaccines is an important factor for increasing vaccination coverage in low-resource settings, contributing to the large share of live attenuated vaccines in the overall market.
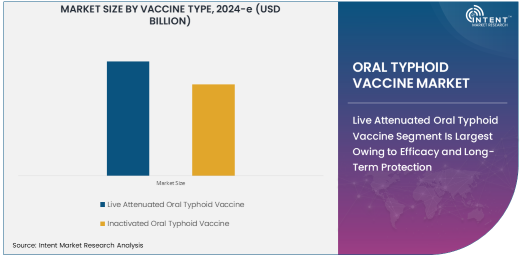
Inactivated Oral Typhoid Vaccine Segment Is Fastest Growing Owing to Enhanced Safety and Stability
The inactivated oral typhoid vaccine segment is the fastest growing in the market, driven by its enhanced safety profile and stability. Unlike live attenuated vaccines, inactivated vaccines contain killed bacteria that cannot cause disease, making them a safer option for certain populations, such as individuals with compromised immune systems or those in sensitive age groups. Inactivated vaccines are also more stable under varying environmental conditions, making them easier to store and transport in areas with limited cold chain infrastructure.
The rising demand for safe and reliable vaccination options, particularly in regions with significant typhoid fever outbreaks, is contributing to the rapid growth of the inactivated oral vaccine segment. Additionally, the increasing adoption of these vaccines in global immunization programs is driving their expansion.
Government Health Programs Segment Is Largest End-Use Industry Owing to Public Health Initiatives
The government health programs segment is the largest end-use industry in the oral typhoid vaccine market, driven by large-scale immunization campaigns aimed at controlling typhoid fever. Governments in endemic regions are increasingly implementing public health initiatives to reduce the incidence of typhoid fever and improve overall public health. Oral typhoid vaccines are a key component of these efforts, offering a cost-effective and easy-to-administer solution for mass vaccination programs.
Governments are also partnering with international health organizations to ensure that vaccines reach vulnerable populations in rural or underserved areas. These initiatives contribute significantly to the large share of government health programs in the oral typhoid vaccine market.
Asia Pacific Is Largest Region Owing to High Incidence of Typhoid Fever and Growing Vaccination Efforts
Asia Pacific is the largest region in the oral typhoid vaccine market, primarily due to the high incidence of typhoid fever in the region and ongoing vaccination efforts to control the disease. Countries such as India, Pakistan, and Bangladesh report a significant number of typhoid fever cases annually, making vaccination a critical part of public health initiatives. The availability of oral typhoid vaccines in these regions is helping to address the growing demand for accessible and effective vaccination solutions.
Furthermore, governments and health organizations in the region are increasingly adopting oral typhoid vaccines as part of mass immunization programs to combat the widespread burden of the disease. This has made Asia Pacific the largest regional market for oral typhoid vaccines.

Competitive Landscape and Key Players
The Oral Typhoid Vaccine Market is competitive, with key players such as Sanofi Pasteur, Bharat Biotech, and GlaxoSmithKline leading the development and distribution of oral typhoid vaccines. These companies are at the forefront of vaccine innovation, focusing on improving vaccine safety, efficacy, and accessibility.
The competitive landscape is marked by ongoing research to develop newer, more effective oral typhoid vaccines, along with efforts to expand access in low-income countries. Strategic collaborations with governments and international health organizations, as well as partnerships with healthcare providers, are key strategies employed by leading players to strengthen their position in the market. As the demand for typhoid vaccination continues to grow, companies in the oral typhoid vaccine space are expected to enhance their product offerings and expand distribution networks to meet the needs of global public health initiatives.
Recent Developments:
- Sanofi Pasteur launched an updated oral typhoid vaccine with improved protection and enhanced storage capabilities.
- Bharat Biotech received regulatory approval for its new oral typhoid vaccine designed for mass immunization campaigns in developing countries.
- Merck & Co., Inc. announced a partnership with global health organizations to distribute oral typhoid vaccines to low-income regions.
- Serum Institute of India Pvt. Ltd. introduced a cost-effective oral typhoid vaccine for use in rural healthcare programs.
- Panacea Biotec expanded its oral typhoid vaccine production capacity to meet the rising global demand for vaccination programs.
List of Leading Companies:
- Sanofi Pasteur
- GlaxoSmithKline
- Bharat Biotech
- Bio-Med Pvt. Ltd.
- Serum Institute of India Pvt. Ltd.
- China National Biotec Group
- Panacea Biotec
- Beijing Tiantan Biological Products
- Indian Immunologicals Ltd.
- Merck & Co., Inc.
- EuBiologics Co., Ltd.
- Novartis AG
- The Institute of Vaccines and Sera
- Crucell (Johnson & Johnson)
- JN International Medical Corporation
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 0.4 Billion |
|
Forecasted Value (2030) |
USD 0.7 Billion |
|
CAGR (2025 – 2030) |
12.2% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Global Oral Typhoid Vaccine Market by Vaccine Type (Live Attenuated Oral Typhoid Vaccine, Inactivated Oral Typhoid Vaccine), by End-Use Industry (Hospitals & Clinics, Government Health Programs, Pharmacies & Retail Healthcare) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Sanofi Pasteur, GlaxoSmithKline, Bharat Biotech, Bio-Med Pvt. Ltd., Serum Institute of India Pvt. Ltd., China National Biotec Group, Beijing Tiantan Biological Products, Indian Immunologicals Ltd., Merck & Co., Inc., EuBiologics Co., Ltd., Novartis AG, The Institute of Vaccines and Sera, JN International Medical Corporation |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Oral Typhoid Vaccine Market, by Vaccine Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Live Attenuated Oral Typhoid Vaccine |
|
4.2. Inactivated Oral Typhoid Vaccine |
|
5. Oral Typhoid Vaccine Market, by End-Use Industry (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Hospitals & Clinics |
|
5.2. Government Health Programs |
|
5.3. Pharmacies & Retail Healthcare |
|
6. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Regional Overview |
|
6.2. North America |
|
6.2.1. Regional Trends & Growth Drivers |
|
6.2.2. Barriers & Challenges |
|
6.2.3. Opportunities |
|
6.2.4. Factor Impact Analysis |
|
6.2.5. Technology Trends |
|
6.2.6. North America Oral Typhoid Vaccine Market, by Vaccine Type |
|
6.2.7. North America Oral Typhoid Vaccine Market, by End-Use Industry |
|
6.2.8. By Country |
|
6.2.8.1. US |
|
6.2.8.1.1. US Oral Typhoid Vaccine Market, by Vaccine Type |
|
6.2.8.1.2. US Oral Typhoid Vaccine Market, by End-Use Industry |
|
6.2.8.2. Canada |
|
6.2.8.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
6.3. Europe |
|
6.4. Asia-Pacific |
|
6.5. Latin America |
|
6.6. Middle East & Africa |
|
7. Competitive Landscape |
|
7.1. Overview of the Key Players |
|
7.2. Competitive Ecosystem |
|
7.2.1. Level of Fragmentation |
|
7.2.2. Market Consolidation |
|
7.2.3. Product Innovation |
|
7.3. Company Share Analysis |
|
7.4. Company Benchmarking Matrix |
|
7.4.1. Strategic Overview |
|
7.4.2. Product Innovations |
|
7.5. Start-up Ecosystem |
|
7.6. Strategic Competitive Insights/ Customer Imperatives |
|
7.7. ESG Matrix/ Sustainability Matrix |
|
7.8. Manufacturing Network |
|
7.8.1. Locations |
|
7.8.2. Supply Chain and Logistics |
|
7.8.3. Product Flexibility/Customization |
|
7.8.4. Digital Transformation and Connectivity |
|
7.8.5. Environmental and Regulatory Compliance |
|
7.9. Technology Readiness Level Matrix |
|
7.10. Technology Maturity Curve |
|
7.11. Buying Criteria |
|
8. Company Profiles |
|
8.1. Sanofi Pasteur |
|
8.1.1. Company Overview |
|
8.1.2. Company Financials |
|
8.1.3. Product/Service Portfolio |
|
8.1.4. Recent Developments |
|
8.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
8.2. GlaxoSmithKline |
|
8.3. Bharat Biotech |
|
8.4. Bio-Med Pvt. Ltd. |
|
8.5. Serum Institute of India Pvt. Ltd. |
|
8.6. China National Biotec Group |
|
8.7. Panacea Biotec |
|
8.8. Beijing Tiantan Biological Products |
|
8.9. Indian Immunologicals Ltd. |
|
8.10. Merck & Co., Inc. |
|
8.11. EuBiologics Co., Ltd. |
|
8.12. Novartis AG |
|
8.13. The Institute of Vaccines and Sera |
|
8.14. Crucell (Johnson & Johnson) |
|
8.15. JN International Medical Corporation |
|
9. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Global Oral Typhoid Vaccine Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Global Oral Typhoid Vaccine Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Global Oral Typhoid Vaccine Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
