As per Intent Market Research, the Generic Pharmaceuticals Contract Manufacturing Market was valued at USD 53.9 billion in 2024-e and will surpass USD 113.8 billion by 2030; growing at a CAGR of 11.3% during 2025 - 2030.
The global generic pharmaceuticals contract manufacturing market plays a pivotal role in the pharmaceutical supply chain, offering cost-effective solutions for drug development and production. With increasing healthcare demands and rising costs of developing new drugs, pharmaceutical companies are increasingly outsourcing their manufacturing needs to contract manufacturers. This trend is especially prominent in the production of generic drugs, which offer a more affordable alternative to branded medications. As the market continues to grow, several key segments emerge, driven by the demand for efficiency, quality, and innovation in drug manufacturing processes.
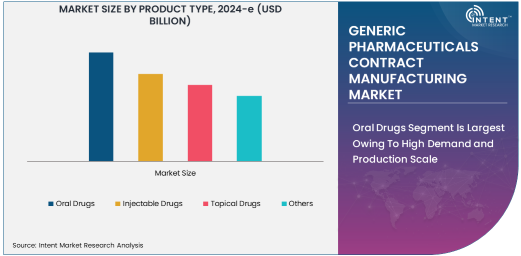
Oral Drugs Segment Is Largest Owing To High Demand and Production Scale
Among various product types, oral drugs dominate the generic pharmaceuticals contract manufacturing market. This segment includes widely consumed formulations such as tablets and capsules, which are often the most cost-effective and scalable production options. Oral medications have gained popularity due to their ease of administration and growing demand for chronic disease management, such as diabetes, cardiovascular diseases, and oncology. The simplicity in formulation and widespread acceptance across demographics further contributes to the large market share of oral drugs.
The growth in the oral drugs subsegment is fueled by increasing patient populations globally and the increasing cost burden of healthcare systems. Contract manufacturers often specialize in the mass production of oral medications, creating efficiencies and cost savings that are crucial for generic drug manufacturers. As the demand for affordable alternatives to branded oral drugs rises, the subsegment is poised to retain its largest share in the market.
Pharmaceutical Companies Drive Market Demand in End-User Industry
In the end-user industry, pharmaceutical companies are the largest segment within the generic pharmaceuticals contract manufacturing market. These companies rely heavily on contract manufacturers to produce a wide range of generic drugs, enabling them to focus on research, marketing, and distribution, while outsourcing production. The increasing need for affordable generics, driven by the rising prevalence of chronic diseases and aging populations, has led pharmaceutical companies to increasingly leverage contract manufacturing organizations (CMOs) to meet the demand.
The preference for outsourcing allows pharmaceutical companies to streamline operations, reduce capital expenditures, and accelerate the time to market for new generic drugs. As generic pharmaceuticals continue to grow in demand, the role of pharmaceutical companies in driving this market forward becomes more significant, particularly in regions with a high demand for low-cost medication.
Manufacturing Services Segment Is Fastest Growing Due to Expanding Production Capacity
In terms of service types, manufacturing services stand out as the fastest-growing segment within the generic pharmaceuticals contract manufacturing market. This growth is primarily driven by the rising demand for generic drug production, as well as the need for specialized manufacturing processes to meet regulatory standards. Contract manufacturers are increasingly focused on providing high-quality, large-scale manufacturing services to meet the growing needs of generic drug companies across a wide range of therapeutic areas.
Manufacturing services are also evolving to incorporate advanced technologies such as automated production lines, improved quality control processes, and compliance with global regulatory standards. This evolution is enabling contract manufacturers to serve a broader range of pharmaceutical companies, including those involved in biologics and other complex drugs. The demand for cost-effective and scalable production continues to push this segment’s growth trajectory.
Tablets Segment Dominates Formulation Type Due to Scalability and Cost Efficiency
Among the various formulation types, tablets lead the generic pharmaceuticals contract manufacturing market. Tablets are a preferred choice due to their versatility, ease of administration, and scalability in mass production. They are suitable for a wide range of therapeutic applications, making them a staple in both over-the-counter and prescription medications. With a focus on reducing costs, manufacturers continue to invest in technology and infrastructure to optimize tablet production, enabling them to meet the growing demand for generics.
The popularity of tablets also stems from their extended shelf life and the ease with which they can be transported and stored. Contract manufacturers that specialize in tablet formulations are benefiting from the large-scale production capabilities required to meet the growing market demand. This dominance is expected to continue as generics for chronic disease management, particularly in cardiovascular and diabetes treatments, rely heavily on tablet forms.
Asia-Pacific Region is Fastest Growing Market for Contract Manufacturing
In terms of geography, the Asia-Pacific region stands as the fastest-growing market for generic pharmaceuticals contract manufacturing. This region benefits from lower labor costs, a robust manufacturing infrastructure, and increasing investments in pharmaceutical manufacturing capabilities. Countries like India and China are key players in the global generic drug production market, and they continue to attract significant foreign investments due to their cost advantages and skilled workforce.
The Asia-Pacific region's rapid growth in contract manufacturing is driven by its ability to meet the increasing global demand for affordable medications, particularly in emerging markets. As the region continues to expand its manufacturing capacity, it is poised to capture an even larger share of the global market, serving both developed and developing economies with high-quality generic drugs.

Leading Companies and Competitive Landscape
The global generic pharmaceuticals contract manufacturing market is highly competitive, with several leading players offering a wide range of services across the product spectrum. Companies such as Lonza Group, WuXi AppTec, Catalent, and Recipharm are at the forefront, providing both manufacturing and specialized services to pharmaceutical companies worldwide. These companies have established themselves as key players by leveraging state-of-the-art technology, adhering to stringent regulatory standards, and expanding their global production capabilities.
The competitive landscape is marked by collaborations, partnerships, and acquisitions, as players seek to enhance their manufacturing capacities and expand their service offerings. The rising demand for cost-effective generic drugs, coupled with increasing regulatory scrutiny, continues to drive innovation in the market. Companies that can efficiently scale production, maintain high-quality standards, and meet regulatory requirements are poised for continued success in this fast-evolving market.
Recent Developments:
- Lonza Group announces the expansion of its manufacturing facilities in the US, with a focus on biologics.
- WuXi AppTec signed a significant contract with a major global pharmaceutical company for comprehensive contract manufacturing services.
- Recipharm AB acquired a contract manufacturing facility in Europe to increase its capacity for injectable drugs.
- Cipla Limited received approval from the FDA for a new generic drug and plans to expand its manufacturing capabilities.
- Sandoz launches a new line of generic injectables to meet rising global demand for affordable medicines.
List of Leading Companies:
- Lonza Group
- WuXi AppTec
- Catalent, Inc.
- Samsung Biologics
- Recipharm AB
- Porton Pharma Solutions
- Cipla Limited
- Aenova Group
- Takeda Pharmaceutical Company
- Boehringer Ingelheim
- Astellas Pharma
- Sandoz International GmbH
- Jubilant Pharma
- Piramal Pharma Solutions
- Alvogen
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 53.9 Billion |
|
Forecasted Value (2030) |
USD 113.8 Billion |
|
CAGR (2025 – 2030) |
11.3% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Generic Pharmaceuticals Contract Manufacturing Market By Product Type (Oral Drugs, Injectable Drugs, Topical Drugs), By End-User Industry (Pharmaceutical Companies, Contract Research Organizations (CROs), Biotechnology Companies, Generic Drug Manufacturers), By Service Type (Manufacturing Services, Packaging Services, Regulatory Support Services, Research and Development Services), By Formulation Type (Tablets, Capsules, Injections, Ointments) |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Lonza Group, WuXi AppTec, Catalent, Inc., Samsung Biologics, Recipharm AB, Porton Pharma Solutions, Cipla Limited, Aenova Group, Takeda Pharmaceutical Company, Boehringer Ingelheim, Astellas Pharma, Sandoz International GmbH, Jubilant Pharma, Piramal Pharma Solutions, Alvogen |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Generic Pharmaceuticals Contract Manufacturing Market, by Product Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Oral Drugs |
|
4.2. Injectable Drugs |
|
4.3. Topical Drugs |
|
4.4. Others |
|
5. Generic Pharmaceuticals Contract Manufacturing Market, by End-User Industry (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Pharmaceutical Companies |
|
5.2. Contract Research Organizations (CROs) |
|
5.3. Biotechnology Companies |
|
5.4. Generic Drug Manufacturers |
|
6. Generic Pharmaceuticals Contract Manufacturing Market, by Service Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Manufacturing Services |
|
6.2. Packaging Services |
|
6.3. Regulatory Support Services |
|
6.4. Research and Development Services |
|
7. Generic Pharmaceuticals Contract Manufacturing Market, by Formulation Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Tablets |
|
7.2. Capsules |
|
7.3. Injections |
|
7.4. Ointments |
|
8. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
8.1. Regional Overview |
|
8.2. North America |
|
8.2.1. Regional Trends & Growth Drivers |
|
8.2.2. Barriers & Challenges |
|
8.2.3. Opportunities |
|
8.2.4. Factor Impact Analysis |
|
8.2.5. Technology Trends |
|
8.2.6. North America Generic Pharmaceuticals Contract Manufacturing Market, by Product Type |
|
8.2.7. North America Generic Pharmaceuticals Contract Manufacturing Market, by End-User Industry |
|
8.2.8. North America Generic Pharmaceuticals Contract Manufacturing Market, by Service Type |
|
8.2.9. North America Generic Pharmaceuticals Contract Manufacturing Market, by Formulation Type |
|
8.2.10. By Country |
|
8.2.10.1. US |
|
8.2.10.1.1. US Generic Pharmaceuticals Contract Manufacturing Market, by Product Type |
|
8.2.10.1.2. US Generic Pharmaceuticals Contract Manufacturing Market, by End-User Industry |
|
8.2.10.1.3. US Generic Pharmaceuticals Contract Manufacturing Market, by Service Type |
|
8.2.10.1.4. US Generic Pharmaceuticals Contract Manufacturing Market, by Formulation Type |
|
8.2.10.2. Canada |
|
8.2.10.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
8.3. Europe |
|
8.4. Asia-Pacific |
|
8.5. Latin America |
|
8.6. Middle East & Africa |
|
9. Competitive Landscape |
|
9.1. Overview of the Key Players |
|
9.2. Competitive Ecosystem |
|
9.2.1. Level of Fragmentation |
|
9.2.2. Market Consolidation |
|
9.2.3. Product Innovation |
|
9.3. Company Share Analysis |
|
9.4. Company Benchmarking Matrix |
|
9.4.1. Strategic Overview |
|
9.4.2. Product Innovations |
|
9.5. Start-up Ecosystem |
|
9.6. Strategic Competitive Insights/ Customer Imperatives |
|
9.7. ESG Matrix/ Sustainability Matrix |
|
9.8. Manufacturing Network |
|
9.8.1. Locations |
|
9.8.2. Supply Chain and Logistics |
|
9.8.3. Product Flexibility/Customization |
|
9.8.4. Digital Transformation and Connectivity |
|
9.8.5. Environmental and Regulatory Compliance |
|
9.9. Technology Readiness Level Matrix |
|
9.10. Technology Maturity Curve |
|
9.11. Buying Criteria |
|
10. Company Profiles |
|
10.1. Lonza Group |
|
10.1.1. Company Overview |
|
10.1.2. Company Financials |
|
10.1.3. Product/Service Portfolio |
|
10.1.4. Recent Developments |
|
10.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
10.2. WuXi AppTec |
|
10.3. Catalent, Inc. |
|
10.4. Samsung Biologics |
|
10.5. Recipharm AB |
|
10.6. Porton Pharma Solutions |
|
10.7. Cipla Limited |
|
10.8. Aenova Group |
|
10.9. Takeda Pharmaceutical Company |
|
10.10. Boehringer Ingelheim |
|
10.11. Astellas Pharma |
|
10.12. Sandoz International GmbH |
|
10.13. Jubilant Pharma |
|
10.14. Piramal Pharma Solutions |
|
10.15. Alvogen |
|
11. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Generic Pharmaceuticals Contract Manufacturing Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Generic Pharmaceuticals Contract Manufacturing Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Generic Pharmaceuticals Contract Manufacturing Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
