As per Intent Market Research, the Anaplastic Astrocytoma Treatment Market was valued at USD 0.6 Billion in 2024-e and will surpass USD 1.1 Billion by 2030; growing at a CAGR of 8.3% during 2025-2030.
The global anaplastic astrocytoma treatment market is expanding as the demand for more effective therapies increases in response to the rising incidence of this aggressive form of brain cancer. Anaplastic astrocytoma, a type of glioma, requires specialized treatment strategies due to its highly malignant nature and resistance to conventional therapies. The market is evolving rapidly, driven by advancements in chemotherapy, radiation therapy, targeted therapies, and immunotherapies, all aimed at improving patient outcomes and extending survival. With significant contributions from leading pharmaceutical companies and continuous research in oncology, the treatment landscape for anaplastic astrocytoma is becoming increasingly sophisticated, providing patients with more tailored and effective solutions. As the market continues to grow, the focus remains on the development of innovative therapies that offer better survival rates and quality of life for patients diagnosed with this rare and aggressive brain cancer.
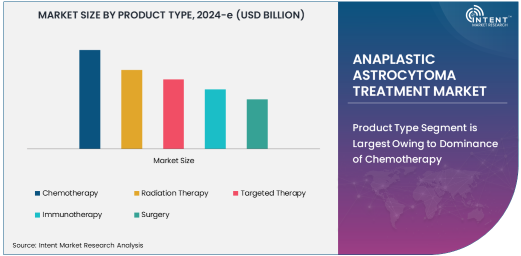
Product Type Segment is Largest Owing to Dominance of Chemotherapy
The Anaplastic Astrocytoma Treatment Market is witnessing significant growth due to the increasing prevalence of anaplastic astrocytoma and the rising demand for effective treatment solutions. Among the various treatment options, chemotherapy remains the largest segment in terms of market share. Chemotherapy, particularly with drugs such as Temozolomide, is the standard treatment for anaplastic astrocytoma due to its ability to slow down tumor growth and improve survival rates. The growing adoption of chemotherapy across healthcare facilities, supported by its proven efficacy in treating brain cancers, makes it the dominant player in the market.
Despite the emergence of new treatment modalities, chemotherapy remains the first-line treatment for most patients diagnosed with anaplastic astrocytoma. The segment continues to receive strong support from ongoing research and development, improving formulations, and combination therapies that enhance its effectiveness. As such, chemotherapy will likely continue to maintain its strong position within the market for the foreseeable future.
Treatment Approach Segment is Fastest Growing Due to Rise in Combination Therapy
The treatment approach segment in the anaplastic astrocytoma treatment market is being led by combination therapy, which is experiencing rapid growth. Combination therapy involves using two or more treatment modalities, such as chemotherapy combined with radiation therapy or targeted therapy. This approach has proven to be more effective than monotherapy in improving outcomes for patients with anaplastic astrocytoma, particularly for those with aggressive or recurrent tumors. The increasing adoption of combination therapies is fueling its growth, as it offers enhanced therapeutic efficacy.
Combination therapy is also gaining traction due to advancements in personalized medicine, where treatments are tailored to the genetic profile of the tumor. This has led to the development of more effective, patient-specific treatment regimens. As clinical trials continue to demonstrate the benefits of these combined approaches, more hospitals and oncologists are adopting this strategy, which is contributing to the rapid expansion of the segment.
Drug Class Segment is Largest Due to Efficacy of Alkylating Agents
In the drug class segment, alkylating agents are the largest subsegment, primarily due to their longstanding use in the treatment of anaplastic astrocytoma. Alkylating agents, particularly Temozolomide, have been shown to be effective in managing gliomas and anaplastic astrocytoma, significantly prolonging survival in patients. Their broad application in both initial and recurrent treatment of the disease ensures their position as the most commonly prescribed class of drugs for brain cancer patients.
These agents work by interfering with the DNA of the cancerous cells, preventing them from dividing and multiplying. As a result, alkylating agents are often used in combination with other therapies to enhance their effectiveness. With continuous advancements in alkylating drug formulations, this drug class is expected to maintain its dominant share of the market, driving the overall growth of the Anaplastic Astrocytoma Treatment Market.
End-User Segment is Largest Owing to Hospital Adoption
The end-user segment of the Anaplastic Astrocytoma Treatment Market is largely driven by hospitals, which account for the largest share of the market. Hospitals, especially those with specialized oncology departments, are the primary settings for the treatment of anaplastic astrocytoma due to the availability of advanced medical equipment, experienced specialists, and multidisciplinary care teams. The presence of comprehensive treatment facilities, including surgery, chemotherapy, and radiation therapy, makes hospitals the preferred destination for patients.
Moreover, hospitals are increasingly investing in cutting-edge diagnostic tools and treatment options to improve patient outcomes. As hospitals continue to be the cornerstone for delivering cancer treatments, their role in the anaplastic astrocytoma treatment market will remain indispensable, ensuring this segment retains its leadership position in the coming years.
Route of Administration Segment is Fastest Growing Due to Rise in Oral Treatments
The route of administration segment is being rapidly shaped by the growing demand for oral medications, making it the fastest-growing subsegment. Oral therapies, such as oral chemotherapy agents like Temozolomide, are gaining popularity due to their convenience and ease of use. Patients prefer oral treatments because they allow for outpatient care, reducing hospital visits and improving their quality of life. As a result, the adoption of oral drugs is increasing, particularly for long-term management of anaplastic astrocytoma.
Oral medications also offer the advantage of better patient compliance compared to intravenous treatments, which require visits to medical facilities. As the development of oral formulations of other cancer therapies advances, this segment is expected to continue its rapid growth, providing a significant shift in how anaplastic astrocytoma is treated.
Distribution Channel Segment is Fastest Growing Due to Surge in Online Pharmacies
The distribution channel segment is being led by online pharmacies, which are emerging as the fastest-growing subsegment in the anaplastic astrocytoma treatment market. The increasing shift toward digital healthcare platforms, accelerated by the COVID-19 pandemic, has led to a surge in online pharmacy usage. Patients can conveniently order their medications from home, which is particularly beneficial for those with chronic conditions like anaplastic astrocytoma, who may require ongoing treatment. Online pharmacies also provide a broader selection of drugs, often with competitive pricing and faster delivery.
This growth in the online pharmacy segment is driven by technological advancements in e-commerce and telemedicine, making access to medications more streamlined and efficient. As patient preferences for home-based treatments increase, the online pharmacy distribution channel is poised to play an increasingly critical role in the market’s development.
Region Segment is Largest in North America Due to Strong Healthcare Infrastructure
North America is the largest region in the Anaplastic Astrocytoma Treatment Market, accounting for the highest market share. The region benefits from a highly developed healthcare infrastructure, a large number of specialized cancer treatment centers, and significant investment in research and development. The presence of major pharmaceutical companies and cutting-edge medical technologies in the United States and Canada also contributes to the region’s dominance in the global market.
Additionally, North America has a high prevalence of brain cancers, including anaplastic astrocytoma, which drives demand for effective treatments. The region's robust healthcare policies and the increasing number of clinical trials aimed at developing new therapies for brain tumors further cement North America’s leadership in the anaplastic astrocytoma treatment space.

Leading Companies and Competitive Landscape
The Anaplastic Astrocytoma Treatment Market is highly competitive, with several global pharmaceutical companies leading the charge in developing and commercializing innovative treatments. Companies such as Merck & Co., Bristol-Myers Squibb, Novartis, and Roche are key players in this market, driving advancements in chemotherapy, radiation therapy, and immunotherapy for anaplastic astrocytoma. These companies have large portfolios of oncology treatments, and many are focusing on combining therapies or developing targeted drugs for better efficacy and minimal side effects.
The competitive landscape is also shaped by increasing collaborations and partnerships between biopharmaceutical companies and research institutions. These partnerships enable the development of novel therapies and clinical trials that aim to improve survival rates for patients with anaplastic astrocytoma. With ongoing research, regulatory approvals, and strategic acquisitions, the competitive dynamics of this market will continue to evolve, fostering further growth and innovation in the coming years.
Recent Developments:
- Merck & Co. announced the FDA approval of its new immunotherapy drug for treating advanced brain cancer, including anaplastic astrocytoma, improving survival rates.
- Bristol-Myers Squibb entered into a partnership with a biotech company for the development of combination therapies targeting glioblastoma and anaplastic astrocytoma.
- Novartis launched a new clinical trial focused on an innovative chemotherapy treatment for anaplastic astrocytoma, aiming to improve patient outcomes.
- Pfizer completed the acquisition of a biotech firm specializing in targeted therapies for brain tumors, including anaplastic astrocytoma, enhancing its portfolio.
- Amgen received regulatory approval in Europe for its new targeted therapy for patients with anaplastic astrocytoma, offering a new treatment option for refractory cases.
List of Leading Companies:
- Merck & Co.
- Bristol-Myers Squibb
- Novartis
- Roche
- Pfizer
- Eli Lilly and Co.
- Amgen
- AbbVie
- AstraZeneca
- Johnson & Johnson
- Bayer
- Celgene Corporation
- Biogen Idec
- Medtronic
- Teva Pharmaceutical Industries
Report Scope:
|
Report Features |
Description |
|
Market Size (2024-e) |
USD 0.6 Billion |
|
Forecasted Value (2030) |
USD 1.1 Billion |
|
CAGR (2025 – 2030) |
8.3% |
|
Base Year for Estimation |
2024-e |
|
Historic Year |
2023 |
|
Forecast Period |
2025 – 2030 |
|
Report Coverage |
Market Forecast, Market Dynamics, Competitive Landscape, Recent Developments |
|
Segments Covered |
Anaplastic Astrocytoma Treatment Market By Product Type (Chemotherapy, Radiation Therapy, Targeted Therapy, Immunotherapy, Surgery), By Treatment Approach (Monotherapy, Combination Therapy), By Drug Class (Alkylating Agents, Antimetabolites, Tyrosine Kinase Inhibitors, Checkpoint Inhibitors, Immunomodulators), By End-User (Hospitals, Specialty Clinics, Research Institutions), By Route of Administration (Oral, Intravenous, Intrathecal), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and By Region |
|
Regional Analysis |
North America (US, Canada, Mexico), Europe (Germany, France, UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, South Korea, Australia, India, and Rest of Asia-Pacific), Latin America (Brazil, Argentina, and Rest of Latin America), Middle East & Africa (Saudi Arabia, UAE, Rest of Middle East & Africa) |
|
Major Companies |
Merck & Co., Bristol-Myers Squibb, Novartis, Roche, Pfizer, Eli Lilly and Co., Amgen, AbbVie, AstraZeneca, Johnson & Johnson, Bayer, Celgene Corporation, Biogen Idec, Medtronic, Teva Pharmaceutical Industries |
|
Customization Scope |
Customization for segments, region/country-level will be provided. Moreover, additional customization can be done based on the requirements |
|
1. Introduction |
|
1.1. Market Definition |
|
1.2. Scope of the Study |
|
1.3. Research Assumptions |
|
1.4. Study Limitations |
|
2. Research Methodology |
|
2.1. Research Approach |
|
2.1.1. Top-Down Method |
|
2.1.2. Bottom-Up Method |
|
2.1.3. Factor Impact Analysis |
|
2.2. Insights & Data Collection Process |
|
2.2.1. Secondary Research |
|
2.2.2. Primary Research |
|
2.3. Data Mining Process |
|
2.3.1. Data Analysis |
|
2.3.2. Data Validation and Revalidation |
|
2.3.3. Data Triangulation |
|
3. Executive Summary |
|
3.1. Major Markets & Segments |
|
3.2. Highest Growing Regions and Respective Countries |
|
3.3. Impact of Growth Drivers & Inhibitors |
|
3.4. Regulatory Overview by Country |
|
4. Anaplastic Astrocytoma Treatment Market, by Product Type (Market Size & Forecast: USD Million, 2023 – 2030) |
|
4.1. Chemotherapy |
|
4.2. Radiation Therapy |
|
4.3. Targeted Therapy |
|
4.4. Immunotherapy |
|
4.5. Surgery |
|
5. Anaplastic Astrocytoma Treatment Market, by Treatment Approach (Market Size & Forecast: USD Million, 2023 – 2030) |
|
5.1. Monotherapy |
|
5.2. Combination Therapy |
|
6. Anaplastic Astrocytoma Treatment Market, by Drug Class (Market Size & Forecast: USD Million, 2023 – 2030) |
|
6.1. Alkylating Agents |
|
6.2. Antimetabolites |
|
6.3. Tyrosine Kinase Inhibitors |
|
6.4. Checkpoint Inhibitors |
|
6.5. Immunomodulators |
|
7. Anaplastic Astrocytoma Treatment Market, by End-User (Market Size & Forecast: USD Million, 2023 – 2030) |
|
7.1. Hospitals |
|
7.2. Specialty Clinics |
|
7.3. Research Institutions |
|
8. Anaplastic Astrocytoma Treatment Market, by Route of Administration (Market Size & Forecast: USD Million, 2023 – 2030) |
|
8.1. Oral |
|
8.2. Intravenous |
|
8.3. Intrathecal |
|
9. Anaplastic Astrocytoma Treatment Market, by Distribution Channel (Market Size & Forecast: USD Million, 2023 – 2030) |
|
9.1. Hospital Pharmacies |
|
9.2. Retail Pharmacies |
|
9.3. Online Pharmacies |
|
10. Regional Analysis (Market Size & Forecast: USD Million, 2023 – 2030) |
|
10.1. Regional Overview |
|
10.2. North America |
|
10.2.1. Regional Trends & Growth Drivers |
|
10.2.2. Barriers & Challenges |
|
10.2.3. Opportunities |
|
10.2.4. Factor Impact Analysis |
|
10.2.5. Technology Trends |
|
10.2.6. North America Anaplastic Astrocytoma Treatment Market, by Product Type |
|
10.2.7. North America Anaplastic Astrocytoma Treatment Market, by Treatment Approach |
|
10.2.8. North America Anaplastic Astrocytoma Treatment Market, by Drug Class |
|
10.2.9. North America Anaplastic Astrocytoma Treatment Market, by End-User |
|
10.2.10. North America Anaplastic Astrocytoma Treatment Market, by Route of Administration |
|
10.2.11. North America Anaplastic Astrocytoma Treatment Market, by Distribution Channel |
|
10.2.12. By Country |
|
10.2.12.1. US |
|
10.2.12.1.1. US Anaplastic Astrocytoma Treatment Market, by Product Type |
|
10.2.12.1.2. US Anaplastic Astrocytoma Treatment Market, by Treatment Approach |
|
10.2.12.1.3. US Anaplastic Astrocytoma Treatment Market, by Drug Class |
|
10.2.12.1.4. US Anaplastic Astrocytoma Treatment Market, by End-User |
|
10.2.12.1.5. US Anaplastic Astrocytoma Treatment Market, by Route of Administration |
|
10.2.12.1.6. US Anaplastic Astrocytoma Treatment Market, by Distribution Channel |
|
10.2.12.2. Canada |
|
10.2.12.3. Mexico |
|
*Similar segmentation will be provided for each region and country |
|
10.3. Europe |
|
10.4. Asia-Pacific |
|
10.5. Latin America |
|
10.6. Middle East & Africa |
|
11. Competitive Landscape |
|
11.1. Overview of the Key Players |
|
11.2. Competitive Ecosystem |
|
11.2.1. Level of Fragmentation |
|
11.2.2. Market Consolidation |
|
11.2.3. Product Innovation |
|
11.3. Company Share Analysis |
|
11.4. Company Benchmarking Matrix |
|
11.4.1. Strategic Overview |
|
11.4.2. Product Innovations |
|
11.5. Start-up Ecosystem |
|
11.6. Strategic Competitive Insights/ Customer Imperatives |
|
11.7. ESG Matrix/ Sustainability Matrix |
|
11.8. Manufacturing Network |
|
11.8.1. Locations |
|
11.8.2. Supply Chain and Logistics |
|
11.8.3. Product Flexibility/Customization |
|
11.8.4. Digital Transformation and Connectivity |
|
11.8.5. Environmental and Regulatory Compliance |
|
11.9. Technology Readiness Level Matrix |
|
11.10. Technology Maturity Curve |
|
11.11. Buying Criteria |
|
12. Company Profiles |
|
12.1. Merck & Co. |
|
12.1.1. Company Overview |
|
12.1.2. Company Financials |
|
12.1.3. Product/Service Portfolio |
|
12.1.4. Recent Developments |
|
12.1.5. IMR Analysis |
|
*Similar information will be provided for other companies |
|
12.2. Bristol-Myers Squibb |
|
12.3. Novartis |
|
12.4. Roche |
|
12.5. Pfizer |
|
12.6. Eli Lilly and Co. |
|
12.7. Amgen |
|
12.8. AbbVie |
|
12.9. AstraZeneca |
|
12.10. Johnson & Johnson |
|
12.11. Bayer |
|
12.12. Celgene Corporation |
|
12.13. Biogen Idec |
|
12.14. Medtronic |
|
12.15. Teva Pharmaceutical Industries |
|
13. Appendix |
A comprehensive market research approach was employed to gather and analyze data on the Anaplastic Astrocytoma Treatment Market. In the process, the analysis was also done to analyze the parent market and relevant adjacencies to measure the impact of them on the Anaplastic Astrocytoma Treatment Market. The research methodology encompassed both secondary and primary research techniques, ensuring the accuracy and credibility of the findings.
.jpg)
Secondary Research
Secondary research involved a thorough review of pertinent industry reports, journals, articles, and publications. Additionally, annual reports, press releases, and investor presentations of industry players were scrutinized to gain insights into their market positioning and strategies.
Primary Research
Primary research involved conducting in-depth interviews with industry experts, stakeholders, and market participants across the E-Waste Management ecosystem. The primary research objectives included:
- Validating findings and assumptions derived from secondary research
- Gathering qualitative and quantitative data on market trends, drivers, and challenges
- Understanding the demand-side dynamics, encompassing end-users, component manufacturers, facility providers, and service providers
- Assessing the supply-side landscape, including technological advancements and recent developments
Market Size Assessment
A combination of top-down and bottom-up approaches was utilized to analyze the overall size of the Anaplastic Astrocytoma Treatment Market. These methods were also employed to assess the size of various subsegments within the market. The market size assessment methodology encompassed the following steps:
- Identification of key industry players and relevant revenues through extensive secondary research
- Determination of the industry's supply chain and market size, in terms of value, through primary and secondary research processes
- Calculation of percentage shares, splits, and breakdowns using secondary sources and verification through primary sources
.jpg)
Data Triangulation
To ensure the accuracy and reliability of the market size, data triangulation was implemented. This involved cross-referencing data from various sources, including demand and supply side factors, market trends, and expert opinions. Additionally, top-down and bottom-up approaches were employed to validate the market size assessment.
NA
